S’pore Receives First Batch Of Pfizer’s Paxlovid Pills For Covid Treatment, Why Isn’t It In M’sia?
 Thirsty for JUICE content? Quench your cravings on our Instagram, TikTok and WhatsApp
Thirsty for JUICE content? Quench your cravings on our Instagram, TikTok and WhatsApp

The first batch of Pfizer’s antiviral oral pill, Paxlovid, arrived in Singapore this week, said Health Minister Ong Ye Kung. The drug is the first oral anti-viral medicine that has been approved by the Health Sciences Authority (HSA) for the treatment of Covid-19 infections.
It will be prescribed and prioritised for those at higher risk of severe Covid-19 illness, Mr Ong said in a Facebook post. He added, “With more treatment options, we are now in a better position to provide good care to Singaporeans infected with Covid-19.”
But when is it coming to Malaysia, and can we trust it?
Pfizer’s drug, Paxlovid, was granted interim authorisation under the HSA’s Pandemic Special Access Route (PSAR) on Jan 31. The authority approved the drug in consultation with its Medicines Advisory Committee.
The drug has to be taken twice daily for five days, and should be given as soon as possible within five days of the onset of Covid-19 symptoms.
The authority said it has reviewed available clinical data for the drug, and found that it could reduce Covid-19 related hospitalisation or death by 88.9 per cent when given within three days from the onset of symptoms.
The efficacy rate was 87.8 per cent when given within five days of symptoms appearing.

This analysis included patients infected with the Delta variant and lab studies have shown that the drug works against the prevailing variants of concern, including Delta and Omicron variants.
Pfizer Singapore country manager Erika Pagani said oral treatments help reduce the severity or onset of illness in Covid-19 patients, and could thus be an important tool in addressing the global impact of the pandemic.
HSA noted that there were generally low numbers of cases with side effects in the clinical study.
Common side effects reported were mild to moderate, such as altered sense of taste, diarrhoea, vomiting, hypertension, muscle pain and chills, it added.
The authority said Paxlovid may also interact with various medications such as medicines for irregular heart rate, migraine, and cholesterol, leading to serious side effects.
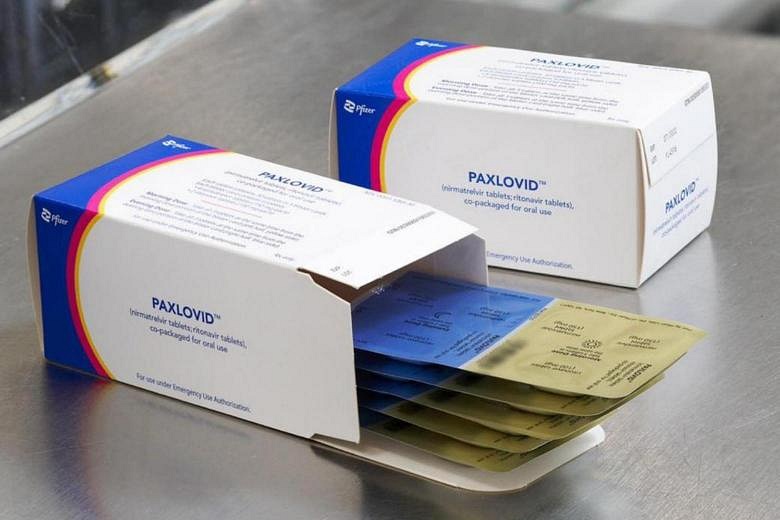
Countries such as South Korea, Britain and Israel have started administering Paxlovid to coronavirus patients from this month, along with these other countries stated by CNA.
It might not be long before we get it here in Malaysia, so maybe hold off on trying outlandish treatments or wild theories on how to “cure” the virus.


 Get Audio+
Get Audio+ Hot FM
Hot FM Kool 101
Kool 101 Eight FM
Eight FM Fly FM
Fly FM Molek FM
Molek FM
